The Delhi High Court dismissed Novartis’s writ petitions, ruling it had waived cross-examination by opting for rebuttal evidence. The Controller’s orders were upheld, with the patent already revoked days earlier.
Read more about Strategic Delay Costs Novartis Its Cross-Examination RightsCategory: Patents
CRI Patentability Affirmed: Madras High Court Rules in Favor of Syngene
Madras HC upholds CRI patentability in Syngene’s case, ruling novel hardware isn’t a prerequisite for protection under Section 3(k).
Read more about CRI Patentability Affirmed: Madras High Court Rules in Favor of SyngeneNon-Disclosure of Prior Art in Hearing Notice Violates Natural Justice, Rules Delhi High Court
In Croda Inc. v. Controller of Patents, the Delhi High Court emphasized procedural fairness and ruled that the non-disclosure of prior art in a hearing notice constitutes a breach of natural justice. The Court remanded the case for fresh evaluation.
Read more about Non-Disclosure of Prior Art in Hearing Notice Violates Natural Justice, Rules Delhi High CourtPharmacyclics Divisional Patent Application Upheld by Calcutta High Court
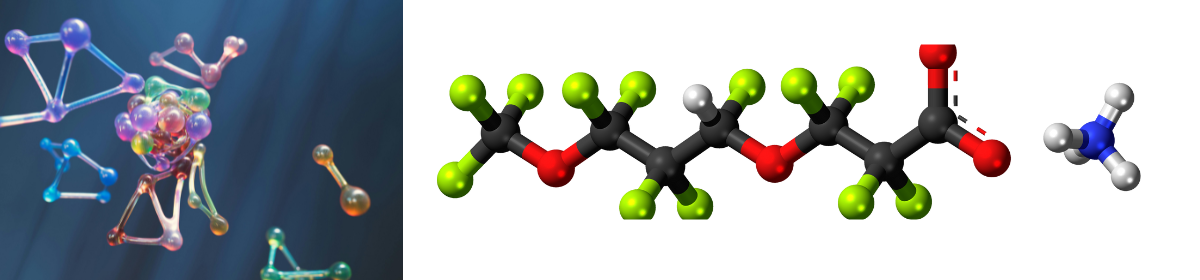
Calcutta High Court upholds Pharmacyclics divisional patent application for ibrutinib-anti-CD20 therapy, interpreting Section 16 of the Patents Act, 1970.
Read more about Pharmacyclics Divisional Patent Application Upheld by Calcutta High CourtFrom Refusal to Reconsideration: A Second Chance for Zhejiang’s Hair Dye Patent
Madras High Court gives Zhejiang’s hair dye patent a new lease on life, quashing an unreasoned refusal and directing a fresh, reasoned hearing.
Read more about From Refusal to Reconsideration: A Second Chance for Zhejiang’s Hair Dye PatentProducts Made During Patent Term are Infringing Even After Expiry
In the case of Boehringer Ingelheim v. Femilab Healthcare, the Himachal Pradesh High Court ruled that interim injunctions granted to prevent patent infringement cannot continue after the patent has expired. The court held that such relief loses effect with the end of the patent term. However, it stated that infringing products made during the patent’s validity may still be restrained from being sold or used after the patent expires.
Read more about Products Made During Patent Term are Infringing Even After ExpiryAmgen’s Lyophilized Peptibody Patent Upheld by Madras High Court
Madras High Court backs Amgen on lyophilized peptibody formulations, rejecting Section 3(d), 3(e) and inventive step objections. Grant directed, with claims narrowed to supported sequences.
Read more about Amgen’s Lyophilized Peptibody Patent Upheld by Madras High CourtOral Insulin Patent Case: Efficacy Not a Requirement Under Section 3(e)
In the case of Oramed Ltd. v. Controller General of Patents & Designs, the Calcutta High Court examined the refusal of a patent for an oral insulin composition. The Court held that inventive step cannot be determined by combining prior art without a clear rationale and stated that therapeutic efficacy is not a requirement under Section 3(e) of the Patents Act. The refusal order was set aside and the matter was remanded for fresh consideration.
Read more about Oral Insulin Patent Case: Efficacy Not a Requirement Under Section 3(e)Cannot raise new ground while refusing patent, rules Delhi High Court
Delhi HC set aside a patent refusal against Proprietect L P, citing violation of natural justice, failure to consider amended claims, and lack of reasoning.
Read more about Cannot raise new ground while refusing patent, rules Delhi High CourtDelhi High Court Patent Injunction: Aquestia Wins Against Automat Industries
Delhi High Court grants patent injunction to Aquestia, stopping Automat from selling Hydromat valves over suspected patent infringement.
Read more about Delhi High Court Patent Injunction: Aquestia Wins Against Automat Industries